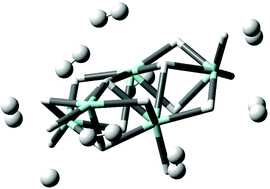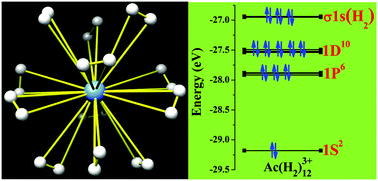
Predicted M(H2)12n+ (M = Ac, Th, Pa, U, La and n = 3, 4) complexes with twenty-four hydrogen atoms bound to the metal ion - Chemical Communications (RSC Publishing)

Sembcorp Industries sinks into the red with S$866m H2 net loss, Companies & Markets - THE BUSINESS TIMES

Jingsha X99 M H2 LGA 2011 3 Motherboard Combo 4* DDR4 RAM Supports Kit Xeon E5 2620 2650 2678 V3 SSD M.2 SATA 3.0 2*PCIE 16X|Motherboards| - AliExpress
Chem 1B Name: Chapter 13 Exercises Exercises #1 1. Consider the following reaction: CO(g) + H2O(g) ⇌

Jingsha X99 M H2 LGA 2011 3 Motherboard Combo 4* DDR4 RAM Supports Kit Xeon E5 2620 2650 2678 V3 SSD M.2 SATA 3.0 2*PCIE 16X|Motherboards| - AliExpress
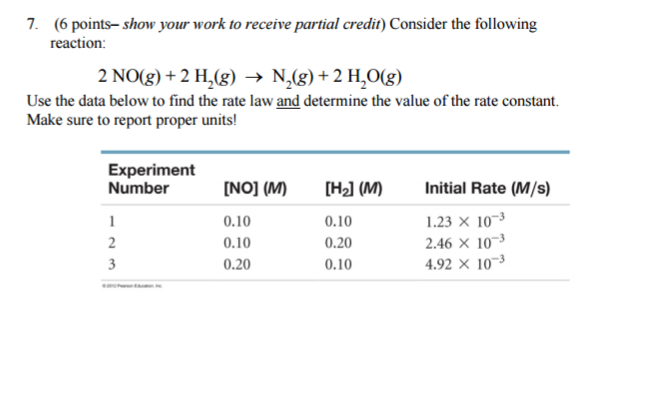
Consider the following reaction: 2 NO(g) + 2 H2(g) → N2(g) + 2 H2O(g) Use the data below to find the rate law and determine the value of the rate constant? Make
The e.m.f. of the cell M |M^n+ (0.02 M) || H+ (1M) H2(g) (1 atm) Pt at 25°C is 0.81 V. - Sarthaks eConnect | Largest Online Education Community
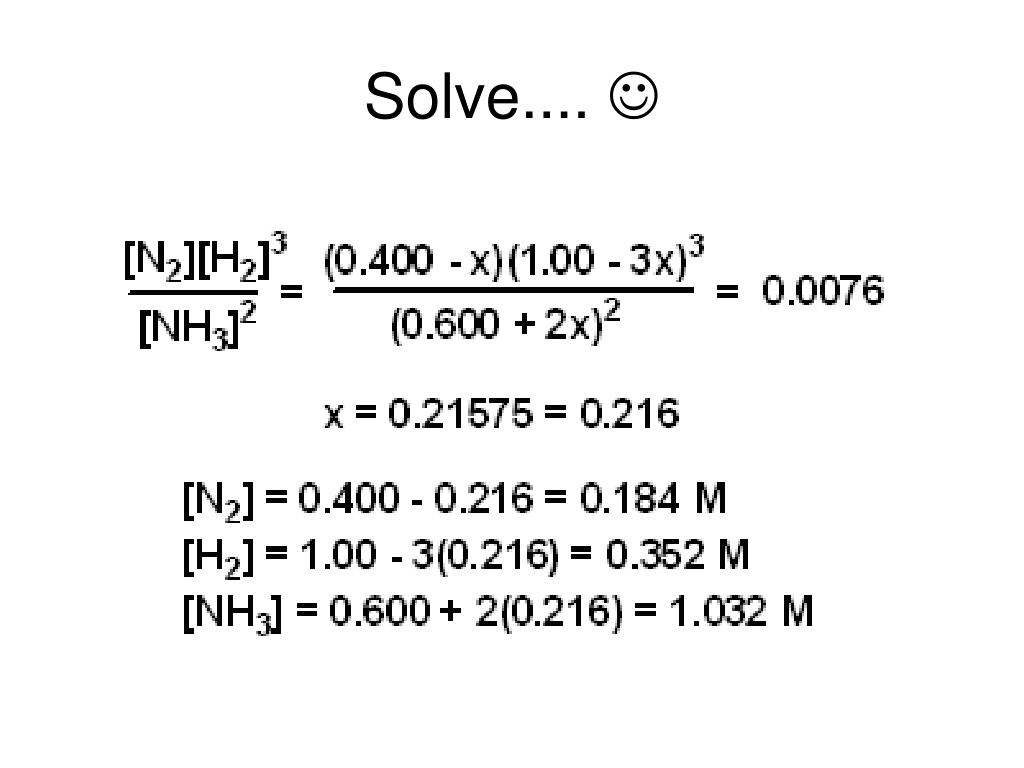
CHM152 Equilibrium Worksheet Key 1 Equilibrium Worksheet Key 1. 2NH3(g) N2(g) + 3H2(g) At 500 K, the following concentrat

For the nitrogen fixation reaction, 3h2(g) + n2(g) 2nh3(g), kc = 6.0 × 10–2 at 500°c. if 0.250 m h2 - Brainly.com

An aqueous solution contains 0.10 M H2 S and 0.20 M HCl . If the equilibrium constants for the formation of HS− from H2S is 1.0×10^−7 and that of S2− from HS−

Jingsha X99 M H2 LGA 2011 3 Motherboard Combo 4* DDR4 RAM Supports Kit Xeon E5 2620 2650 2678 V3 SSD M.2 SATA 3.0 2*PCIE 16X|Motherboards| - AliExpress